
ABSTRACT:
In this article, we will delve into the fascinating world of inorganic chemical reactions in chemistry. By exploring the intricate mechanisms and properties of these reactions, we aim to gain a deeper understanding of the fundamental principles that govern chemical processes. Join us as we embark on this captivating journey of discovery, where we will unveil the secrets held within the realm of inorganic chemistry and shed light upon its various applications in the world around us.
INTRODUCTION OF INORGANIC CHEMICAL REACTIONS:
Chemical reactions are the fundamental processes that drive the transformations of matter. They occur when substances interact with one another, resulting in the formation of new substances with different properties. Understanding the various types of chemical reactions is crucial for scientists and students alike, as it provides insights into the behavior of matter and enables the development of new materials and technologies. In this article, we will explore some of the most common types of chemical reactions, highlighting their characteristics and providing references for further reading.
TYPES OF INORGANIC CHEMICAL REACTIONS:
1. COMBINATION REACTIONS:
Combination reactions, also known as synthesis reactions, occur when two or more substances combine to form a single product. These reactions represented by the general equation: A + B → AB. An example of a combination reaction is the synthesis of water from hydrogen and oxygen: 2H₂ + O₂ → 2H₂O. Combination reactions are essential in the formation of compounds and widely used in industries such as pharmaceuticals and materials science.

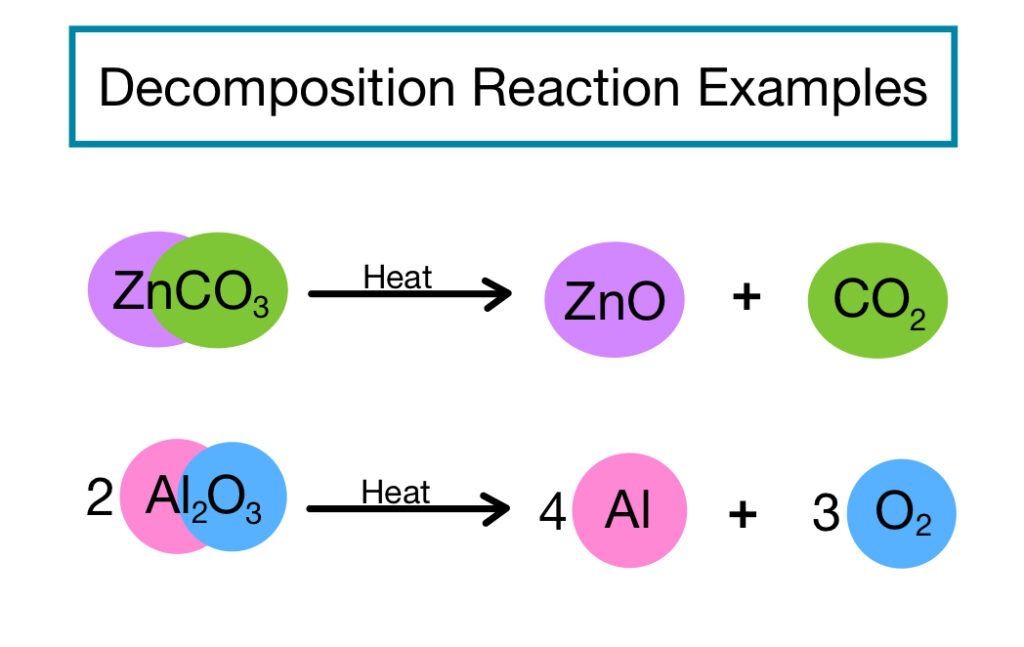
2. DECOMPOSITION REACTIONS:
Decomposition reactions involve the breakdown of a single compound into two or more simpler substances. The general equation for decomposition reactions is: AB → A + B. An example of a decomposition reaction is the breakdown of hydrogen peroxide into water and oxygen gas. 2H₂O₂ → 2H₂O + O₂. Decomposition reactions are essential in nature, such as the decay of organic matter. They also utilized in various industrial processes.
3. SINGLE REPLACEMENT REACTIONS:
Single replacement reactions, also known as displacement reactions, occur when one element replaces another element in a compound. The general equation for single replacement reactions is: A + BC → AC + B. An example of a single replacement reaction is the reaction between zinc and hydrochloric acid: Zn + 2HCl → ZnCl₂ + H₂. Single replacement reactions are crucial in understanding the reactivity of elements. They widely used in the production of metals and in electrochemical processes.

4. DOUBLE REPLACEMENT REACTIONS:
Double replacement reactions occur when the cations and anions of two different compounds switch places, resulting in the formation of two new compounds. The general equation for double replacement reactions is: AB + CD → AD + CB. An example of a double replacement reaction is the reaction between silver nitrate and sodium chloride, resulting in the formation of silver chloride and sodium nitrate: AgNO₃ + NaCl → AgCl + NaNO₃. Double replacement reactions are essential in the formation of precipitates, acid-base neutralization, and the production of salts.

CONCLUSION OF INORGANIC CHEMICAL REACTIONS:
Chemical reactions are the driving force behind countless natural and industrial processes. By understanding the different types of reactions, scientists can predict and control the behavior of matter, leading to advancements in various fields. This article has provided a brief overview of some common types of chemical reactions, including combination, decomposition, single replacement, and double replacement reactions. For further exploration, the provided references offer in-depth information on each reaction type, allowing readers to delve deeper into the fascinating world of chemistry.
REFERENCES:
Chemical Reactions: Synthesis Reactions.” Chem.libretexts.org, Libretexts, 21 July 2016. https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-The_Central_Science(Brown_et_al.)/03%3A_Stoichiometry-_Chemical_Formulas_and_Equations
Chemical Reactions: Decomposition Reactions. Chem.libretexts.org, Libretexts, 21 July 2016, https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-The_Central_Science(Brown_et_al.)/03._Stoichiometry%3A_Calculations_with_Chemical_Formulas_and_Equations/3.4%3A_Decomposition_Reactions.
Chemical Reactions: Single Replacement Reactions.” Chem.libretexts.org, Libretexts, 21 July 2016, http://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-The_Central_Science(Brown_et_al.)/03._Stoichiometry%3A_Calculations_with_Chemical_Formulas_and_Equations/3.6%3A_Single_Replacement_Reactions
Chemical Reactions: Double Replacement Reactions.” Chem.libretexts.org, Libretexts, 21 July 2016, http://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-The_Central_Science(Brown_et_al.)/03._Stoichiometry%3A_Calculations_with_Chemical_Formulas_and_Equations/3.7%3A_Double_Replacement_Reactions


